
Special care must be taken when working with dry ice to avoid frostbite. At atmospheric pressures found on Earth, dry ice undergoes sublimation at −109.3 ☏ ( −78.5 ☌). A white, opaque solid, it isĪlso widely used as a cryogenic agent in industry to reduce bacteria growth and maintain low temperatures. The sublimation point of a substance is the temperature at which it changes directly from a solid to a vapor.ĭry ice (solid CO 2), which rapidly undergoes sublimation from solid to vapor at room temperatures, is often used to create fog on stage and movie sets. Substances that behave in this way are said to sublime. Instead, it changes directly into a vapor. When solid iodine is heated, it does not melt. They may pass directly from the solid state to the gaseous state. Some materials behave differently from water when they are heated. The freezing point is the same as the melting point of the solid. The freezing point of a liquid is the temperature at which the liquid changes to a solid. At this point, the liquid becomes frozen. Under the proper conditions, at some point, the liquid cools sufficiently to change to a solid. It is the same as the boiling point of the liquid. The condensation point is the temperature at which a gas or vapor changes to a liquid. 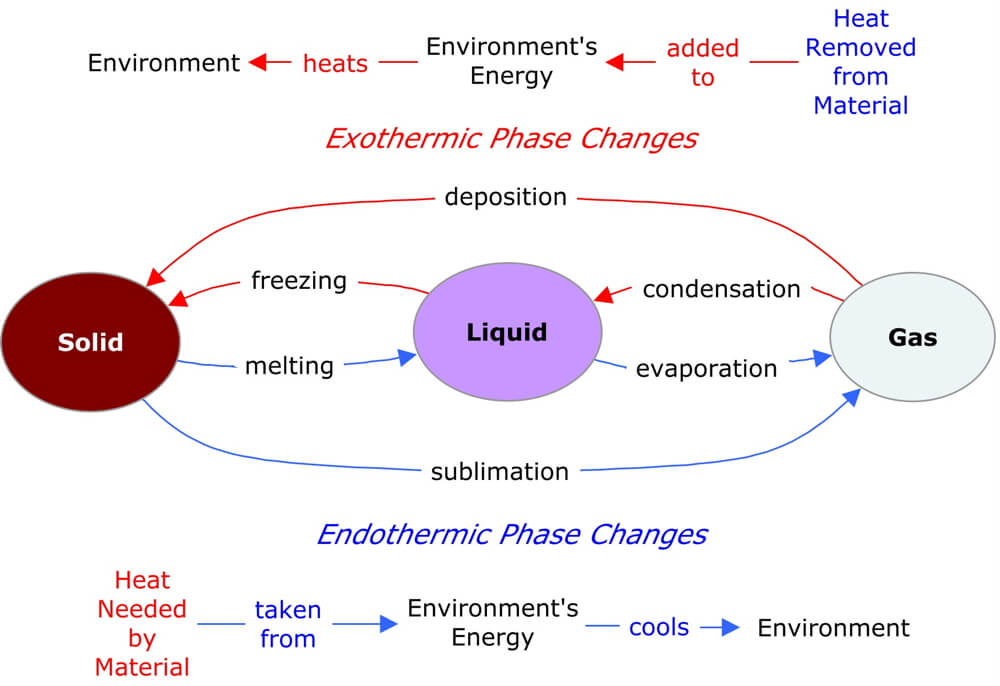
The steam is said to condense to a liquid. When that happens, the water reverts to a liquid. 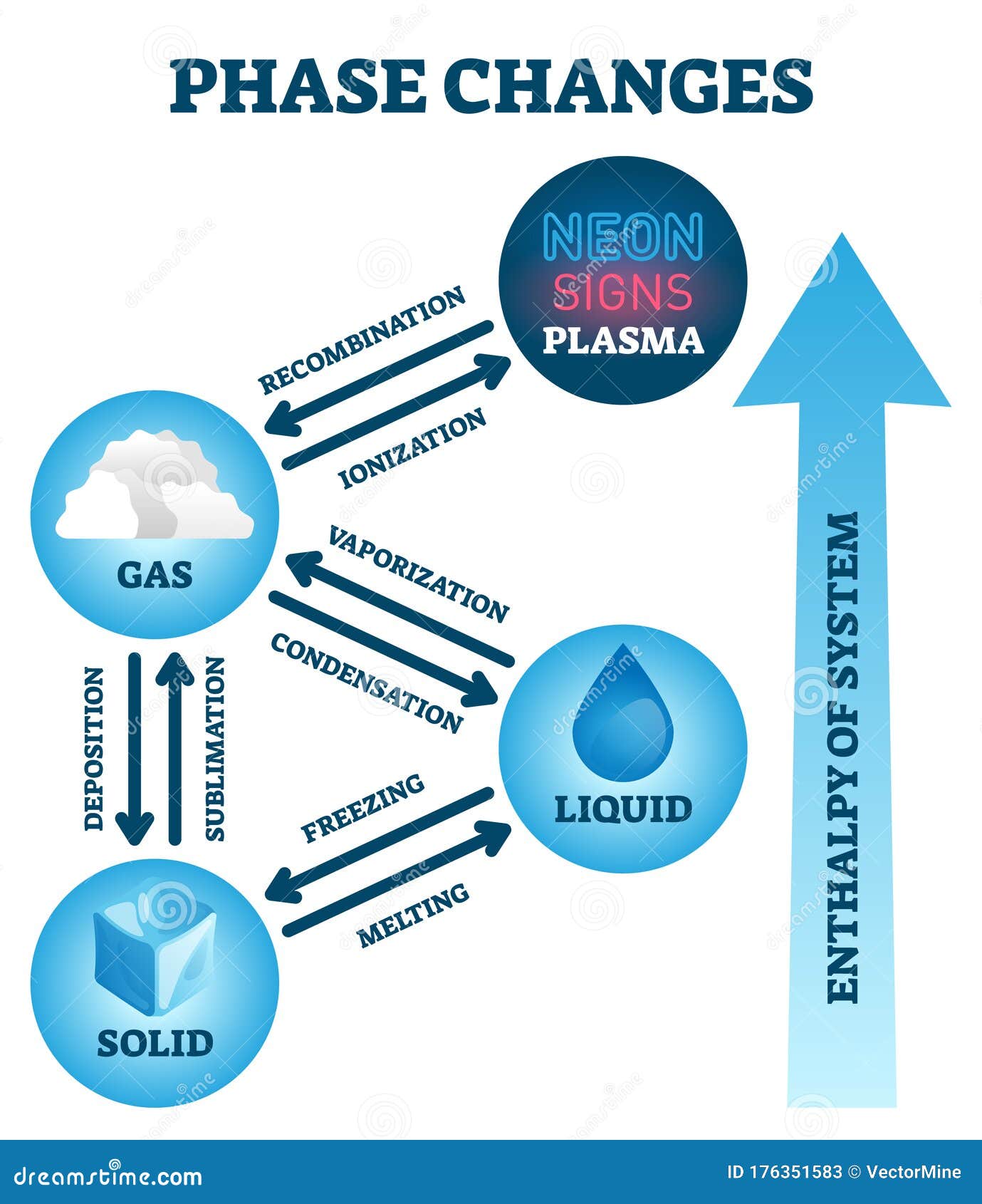
Suppose the steam in this example is cooled below 212 ☏ (100 ☌). If the steam formed in this process is heated further, its temperature continues to increase.Ĭhanges of state occur also when a material is cooled. The lower the pressure (e.g., lower pressures found at altitude) the lower the boiling point. The temperature is a function of atmospheric pressure. The temperature at which a liquid changes to a gas is called its boiling point. At a temperature of 212 ☏ (100 ☌), the water molecules are moving fast enough to change to a vapor, called steam. The temperature of the liquid water increases from 32 ☏ (0 ☌) to 212 ☏ (100 ☌). The increase in speed with which they move is measured as an increase in temperature. If additional heat is added to the liquid water, water molecules move even faster. The melting point is the temperature at which a solid changes to a liquid. The point at which this occurs is the melting point. 
At some point, they vibrate rapidly enough to break the lattice array and move freely in a liquid state. As heat is added to the ice, the molecules begin vibrate more rapidly. The molecules of water in the ice are vibrating in a crystalline array. The term vapor is used to describe the gaseous state of a substance that is normally a liquid at room temperature. Eventually, they are moving fast enough to change to the gaseous, or vapor, state. When water is heated, molecules begin to move more rapidly. The state in which matter occurs can be changed by changing the energy state of the matter or the system surrounding matter that has the capacity to come into equilibrium with that system. The particles in a gas are at an even higher energy level and move most rapidly. The particles in a liquid are at a higher energy level and move more rapidly. The particles in a solid contain relatively little energy and move slowly. One fundamental way in which these three states differ from each other is the energy of the particles of which they are made. Matter commonly exists in one of three forms, or states: solid, liquid, or gas. A change of state is usually accompanied by a change in temperature and/or pressure. For example, when water is heated, it changes from a liquid to a gas -when cooled water will eventually freeze into a solid: ice. A change of state occurs when matter is converted from one physical state to another.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
